Kanev, I., DeHaai, K.A. Van Dyke, A. Z., Sanmann, J.N., Hess, M.M., Starr, L.J., Dave, B.J., and Sanger, W.G. University of Nebraska Medical Center, Munroe-Meyer Institute, Human Genetics Laboratory Omaha, Nebraska, USA. E-mail: [email protected]
Abstract-This paper presents information and raises questions about some amazing structural and functional similarities between human chromosomes and two electrical transformers, the conventional transformer and the Tesla coil. In its tightly coiled and highly repetitive segments of DNA, known as heterochromatin, chromosomes are similar to the conventional transformer. In its loosely coiled segments of DNA, known as euchromatin, chromosomes are similar to a Tesla coil transformer. Some of the defects observed in conventional transformers, including gaps, breaks, and fusions, are similar to events that occur in heterochromatin-rich regions of the chromosomes. Some of the effects that occur in a Tesla coil transformer, including wireless transportation of power and signals, are similar to events which are well expressed and clearly visible in the chromosome regions rich in heterochromatin. The questions are: Are these similarities coincidental or real? Is it possible for chromosomes to operate with energy (or power) like electrical transformers? Are the heterochromatin areas really critical and responsible for formation of gaps, breaks, fusion and other types of chromosomal abnormalities? If the answers to these questions are positive, then chromosomes should be described not only as vehicles for carrying genes and inheritance but also as transformers operating with energy. Furthermore, if true, it will change one of the oldest and main principles in cytogenetics, specifically that the heterochromatin is not important in formation of genetically based diseases and syndromes. We hypothesize that if rich in heterochromatin areas affect the transmission of energy within chromosomes causing disturbances, then these disturbances might cause gaps, breaks, fusion and other kind of damaging of the DNA molecules, and these damages could be a main mechanism in the formation of chromosome abnormalities and various cancers, infertility, pregnancy loss, and syndromes (e.g. Down and Turner syndromes) with chromosomal etiologies.
HISTORY
The history of chromosomes and electrical transformers developed simultaneously in two different fields of science, biology and physics. In biology, the history of human chromosomes can be traced back to the 19th century beginning with biologist Walther Flemming [1], who named the intensely staining intranuclear structures of cells, chromatin (colored body). A few years later it was renamed chromosome by biologist Heinrich Wilhelm Gottfried von Waldeyer Hartz [2]. Thomas Morgan and co-workers [3] discovered that genes are carried on chromosomes and are the mechanical basis of heredity. This discovery formed the basis of the modern science of genetics.
In physics, during the same period of time (circa 1880-1910) two systems for distributing electrical power were developed. The first system, known as direct current (DC), was developed by Thomas Edison. This system was designed for low voltage, making it safe but difficult in voltage conversion. The second system, known as alternating current (AC), was developed by Nikola Tesla. His idea was to generate AC power of any convenient voltage and step up the voltage for transmission. The step up and step down voltage conversion was based on the use of a transformer, a device that converted one AC voltage to another AC voltage at the same frequency. The first commercial transformer was built in 1885 by William Stanley [4]. His transformer known today as a conventional transformer was built from coils of wire wrapped around a ferromagnetic core. In 1891, Tesla [5] invented a new type of transformer, known today as a Tesla coil. It is an air-core, dual-tuned, resonant transformer for generating very high voltage and high frequency. The main purpose of his apparatus was to convert and supply electrical energy in a form suited for the production of certain novel electrical phenomena. This novel phenomena was demonstrated by Tesla in presence of Albert Einstein, Mark Twain, and other prominent scientists and intellectuals in his time.
Interestingly, the human chromosome and the electrical transformer show amazing similarities in some of their constructions, effects, and defects.
CONSTRUCTION
Chromosomes and transformers are amazingly similar in their construction, sharing a similar principal of organization. Both are constructed of long, coiled, string-shaped material. The building material of a Tesla coil and conventional transformer is metal wire with specific requirements for its conductivity, size, position, and number of coils. In a conventional transformer, the coils are very tightly coupled over the iron core (Fig. 1) and voltage gain is determined by the number of turns in the coils. This works well at normal voltages but is inconvenient for high voltage operations. In a Tesla coil transformer, the windings are loosely coupled, with large air gaps between the windings (Fig. 2). As a result of this construction, the primary and secondary coils typically share only 10-20% of their respective magnetic fields.
In chromosomes, the building material is DNA, known as chromatin, which is coiled and packed with the support of histone and non-histone proteins. Chromatin is divided into two types, euchromatin and neterochromatin (Fig. 3). Euchromatin is loosely coiled, similar to the windings of a Tesla coil transformer. Heterochromatin is tightly coiled, similar to the windings of conventional transformer.
According to Anthony Annunziato [6], the human body contains about 50 trillion diploid cells, each containing about 2 meters of DNA within the nucleus. From this number, he estimated that the human body contains 100 trillion meters of DNA, which is enough DNA to go from Earth to the Sun and back more than 300 times. Despite years of speculation, Woodard [7] explained how two meters of DNA is packed and stored in each diploid cell nucleus, which is only 10 to 30 microns in diameter. This makes the human chromosomes one of the most coiled structures found in living and non-living material.
Tesla invented his transformer about 60 years before James Watson and Francis Crick [8] discovered the double helix structure of DNA and about 90 years before the first official idiograms and illustrations of the shape of human chromosomes were published in the first edition of ISCN [9].Yet, as shown in Figures 2 and 3, his transformer is amazingly similar in shape and in principal construction to human chromosome 21.
Fig. 1
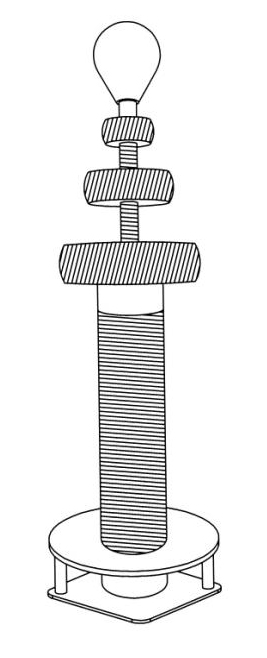
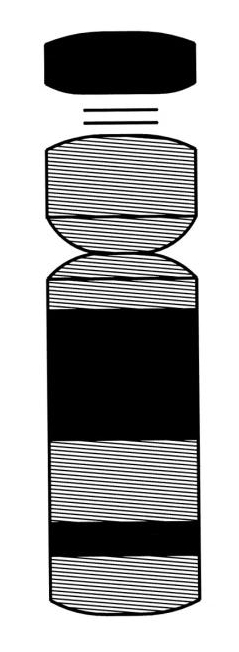
Figures 1, 2 and 3. Graphic illustration of a conventional transformer of Stanley (1), a Tesla coil transformer (2), and a human chromosome 21 (3) enlarged to match the size of Fig.2. In its tightly coiled and highly repetitive rich in heterochromatin areas (illustrated in black) chromosome 21 of Fig. 3 is similar in construction to the tightly coiled windings of the conventional transformer of Fig. 1. In its loosely coiled areas (illustrated with lines) human chromosome 21 is similar in construction to the loosely coils of the Tesla coil transformer of Fig. 2.
The reasoning for the construction of both conventional and Tesla coil transformers is well understood. However, the purpose of the shape and construction of chromosomes remains a speculation. For many years it was believed that the only purpose of tightly coiled DNA was to fit chromosomes into the small cell nucleus. Recently it was discovered that the coiling and uncoiling of DNA play important roles in gene expression and regulation. A new branch of genetics called Epigenetics was created to study the heritable changes in gene expression or cellular
phenotype caused by mechanisms other than changes in the underlying DNA sequence. We hypothesize that perhaps a chromosome’s coils play another important role connected with energy or power.
EFFECTS
In both a conventional and a Tesla coil transformer, effects are known to occur after being charged with energy. Among many of its effects, a Tesla coil is well known for its unique ability to act as a magnifying transmitter, in which it discharges high frequency energy that is visible and safe under specific conditions. Additionally, a Tesla coil possesses the phenomenal ability to transmit signals and power from one transformer to another and from one place to another without using wires. This phenomenon was demonstrated by Tesla in many different ways, including lighting an electrical bulb in his hand without direct connection to the Tesla coil transformer sitting several meters away.
Chromosomes could be charged. This is known from 1891 when Ruzicka [10] used silver staining to study structures (nucleolar organizing areas, NORS) inside of the cell nucleus. In 1928, Emil Heitz [11] discovered that in packed chromosomes NORS are located on the p-arm of the acrocentric chromosomes in regions known as satellites and stalks. In 1933, Harriet Creighton and Barbara McClintock [12] identified a phenomenon known as satellite association. These associations occur when charged satellites of two or more chromosomes are attracted. In 1983, Verma et al. [13] discovered that NORS are stained with silver and formed associations only if they are actively charged. In a long and extensive literature search, we failed to find information about the origin and type of energy which is responsible for charging satellites and leading to satellite associations. In 12 years of focused studies with human chromosomes, we have observed satellite associations and centric fusions using fluorescence in situ hybridization (FISH; Fig 4 and 5) and other methods. However, the nature and the origin of the energy responsible for these charges are not well understood.
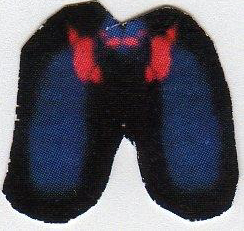

Figures 4 and 5. FISH photographs of two pairs of chromosomes 21. Chromosomes photographed in Fig.4 possess dots of red labeled particles located between the more robustly red labeled satellites. These findings are amazingly similar to the lightning particles which occurred between the two charged Tesla coil transformers shown in Fig. 6. Chromosomes photographed in Fig. 5 showed fused satellites and arched forebodies as a result of electrical (electromagnetic? or electrostatic?) forces which are suggested to be responsible for the fusing of satellites.

Two Tesla coil transformers exchanging energy and signals (From Tesla coil images)
DEFECTS
In the conventional transformer, one type of defect can occur when the winding of the coils is too tight and accumulates over the iron core. In such a construction, according to the laws of physics, the primary and secondary windings share 80-90% of their magnetic fields, and the insulation between the two sets of windings is easily broken down, causing electrical sparks, cracks, splits, bursts, breaks, and fusions.
In human chromosomes, similar gaps, breaks, fusions, and other defects were observed as early as 1915 when Morgan et al. [3] used X-rays to cause mutations in the chromosomes of the fruit fly Drosophila melanogaster. Using the same methodCreighton and McClintock [12] observed gaps, breaks, associations, translocations, and other defects in chromosomes of corn (maize) exposed to X-rays. They noticed that defects occurred in the areas which are rich in heterochromatin forming knob like structures at the end of chromosomes. In human chromosomes, areas rich in heterochromatin are located in many places including in the centromeres and telomeres of all chromosomes, in the satellites of acrocentric chromosomes 13, 14, 15, 21, and 22, and in the variable qh regions of chromosomes 1, 9, 16 and Y. Ferguson Smith and Handmaker [14] studied breaks, fusions, and other defects in human chromosomes rich in heterochromatin and suggested that they play an important role in the formation of some genetically based conditions and diseases. Their suggestions were rejected because the mechanism of these defects is not understood.
Like the previous studies with chromosomes exposed to X-ray [3, 12] our studies indicate that brakes, fusions and other defects occur in chromosomes more often and mainly in locations rich in tightly coiled heterochromatin. In fig. 7 and 8 are pictures of human chromosome number 1 after exposed to toxic chemicals DEB (1,2,3,4-Depoxibutane) and MMC (Mitomycin C) which are used in clinical genetics for so called „breakage study“.


Figures 7 and 8. Photographs of chromosome 1 following exposure to DEB and MMC. Breaks (7) and fusions (8) are found in the heterochromatin-rich qh locations.
The mechanism for the formation of these breaks and fusions is not currently understood. It is unknown why exposure to the same amount and type of X-rays, toxic chemicals or other ionizing on non-ionizing radiation causes some chromosomes to remain intact while others are broken, or fused mainly on the locations which are rich with heterochromatin. Our suggestion is perhaps this occurs because when charged with energy chromosomes react in two different ways. When exposed to energy, chromosomes with enlarged segments of tightly coiled heterochromatin show gaps, breaks, and other defects, which are similar to those defects observed in the tightly coiled windings of the conventional transformer. Alternatively, like Tesla coil transformers, chromosomes with less heterochromatin are more likely to remain stable.
APPLICATION
If our suggestions described above are real and the human chromosome behaves differently in respect to the amount of heterochromatin, it could have widespread impact on the understanding of the mechanism of some of the most common and dangerous diseases, syndromes, and conditions in human history, including cancer, mental retardation, infertility, and pregnancy loss.
For example, cancer (including many types of solid tumors and blood cancers) has existed for many years with an unknown mechanism of origin and formation. Avery Sandberg [15] and other scientists have suggested a positive correlation between the amount of tightly coiled, highly repetitive, heterochromatic regions of chromosomes and some types of cancer. Like Sandberg’s studies our experience in chromosomal cancer studies [16] has indicated a greater occurrence of large heterochromatic regions in the chromosomes of patients with some types of solid tumors and blood cancers. The causative mechanism of the heterochromatin in the formation, diagnosis, prognosis, and treatment of cancer could be better understood if our suggestions are correct and the work of chromosomes is really dependable on energy.
Mental retardation, including those caused from the absence of a whole chromosome (monosomy; e.g., monosomy X or Turner syndrome) or from the addition of a whole chromosome (trisomy; e.g., trisomy 21 or Down syndrome), has existed for many years without a known mechanism of formation. Ohno et al.[17] suggested that there is correlation between the heterochromatin regions of chromosomes and the formation of trisomy and other anomalies and syndromes in humans, but the mechanism of this correlation is unknown, not understood, and has raised considerable doubt by Felix Cruz, Park Gerald [18] and others. In our studies involving families with Down syndrome [19, 20], we have experienced the same correlations. Based on these studies, we suggest that the mechanism of formation of Down, Turner, and other syndromes could be better understood, diagnosed, and treated if the similarities between chromosomes and electric transformers are more than coincidental in nature.
Infertility and pregnancy loss have been recognized for centuries, but the mechanism of formation is not understood. Anuradha et al.[21] suggested that there is correlation between the enlarged areas of heterochromatin-rich satellites, satellite associations in chromosomes, and recurrent abortion, but the mechanism of this correlation is unknown. In our studies with chromosomal abnormalities of sex chromosomes (X and Y) [22], we have experienced the same correlation and problems. Now we believe that there is a possibility for a better understanding of the problems with infertility and pregnancy loss if chromosomes can operate as a magnifying transmitter, discharging energy and transmitting signals and power from one chromosome to another, in a way that is similar to the Tesla coil transformer.
The amazing similarities between human chromosomes, a Tesla coil, and the conventional transformer described in this paper have gone unrecognized for more than 120 years due to a communication gap between the scientific disciplines of physics and biology. In part this may be due to the different methodologies and different purposes in the respective fields, but it may also be due to unshared professional knowledge, skills, and experience across disciplines. To determine if the similarities between the Tesla coil and chromosomes are real or coincidental collaborations among scientists and professionals in the field of genetics, molecular biology, physics, chemistry, modern optics, and computer technology are needed for the observation and investigation of DNA structure and function.
Acknowledgments: This study was supported by internal cytogenetics research and development funds from Human Genetics Laboratory, Munroe-Meyer Institute, University of Nebraska Medical Center. Thanks are given to our colleagues Dianna Zaleski, Siri Huston, Kelly Novak, and Li Zeng for their professional support during the course of this study. We are grateful to Dr. Richard Lutz for his critical opinion, to Mrs. Vassilka Dimitrova and to Mrs. Kristi Horeis for their excellent technical support.
REFERENCES
[I] W. Flemming, „Zur Kenntniss der Zelle und ihrer Theilungs-Erscheinungen,“ In: Schriften des Naturwissenschaftlichen Vereinsfiir Schleswig-Holstein, 1878, vol. 3 pp.23-27.
[2] H. W. G. von Waldeyer, „Uber Karyokinese und ihre Beziehungen zu den Befruchtungsvorgangen,“ Archiv fur
mikroskopische Anatomie und Entwicklungsmechanik, 1888, vol. 32, pp 1-122.
[3] T. H. Morgan, A. H. Sturtevant, H. J. Muller and C. B. Bridges, „The Mechanism ofMendelian Heredity“ 1915, New York, Henry Holt Publisher, pp. 1-262.
[4] W. Stanley/Tnduction coil (The practical coil circuits), invented 1885,U. S. Patent 349611, dated 21 May, 1886.
[5] N. Tesla,“System of Electric Lighting,“ U.S. Patent 454,622, June 23, 1891.
[6] A. T. Annunziato, „DNA packaging: Nucleosomes and chromatin,“ Nature Education, 2008, 1(1) pp.28-36.
[7] C. L. Woodcock, „A milestone in the odyssey of higher-order chromatin structure,“ Nature Structural and Molecular Biology, 2005, 12, 639-640.
[8] J. D. Watson and F. H. Crick, „A structure for deoxyribose nucleic acids,“ Nature, 1953, vol. 171(4356), pp. 737-738.
[9] ISCN: „An International System for Human Cytogenetic Nomenclature, “ Birth defects: Original Article Series, 1978, vol. 14(8), The National Foundation, New York; also in Cytogenetic Cell Gent, vol.21, pp. 309-404.
[10] V. Ruzicka, Zur geschichte und kenntinis der feineren structur der nucleolen centralen nervzellen. Anat Anz, 1891, vol.16, pp. 557-563.
[11] E. Heitz, Das Heterochromatin des Moose.L, Jahre Wiss. Bot, 1928, vol. 69, pp. 762-818.
[12] H. B. Creighton and B. McClintock, „A Correlation of Cytological and Genetical Crossing-Over in Zea Mays“. Proceedings of the National Academy of Sciences of the United States of America, 1931, vol. 8, pp. 492-497. [13] R. S. Verma, J. Rodriguez, J. V. Shah, H. Dosik, Preferential association of nucleolar organizing human chromosomes as revealed by silver staining technique at mitosis. Mol. Gen. Genet, 1983, vol. 190, pp. 325-354. [14] M. A. Ferguson Smith and S. D. Handmaker, „Observation on the satellite human chromosomes,“ Lancet, 1961, vol. 1, pp. 638-640.
[15] A. A. Sandberg, „The chromosomes in Human Cancer and leukemia,,, 2nd Edition, New York, Elsevier Publisher, pp. 1-280.
[16] I. Kanev, D. Pickering, M. Wiggins, A. Milev, A. Kril, B. Dave and W. Sanger, „CCND1 Duplication in a breast cancer patient with thyroid and respiratory disease,“ The Journal of the Association of Genetic Technologists, 2007, vol. 33(3) p. 143.
[17] S. Ohno, J. M. Trujillo, W. B. Kaplan, „Nucleolus organizer in the causation of chromosomal anomalies in man,“ 1961, Lancet, 2, pp. 123-126.
[18] F. F. de la Cruz and P. S. Gerlad, „Trisomy 21 (Down syndrome), 1981, University Park Press, pp.1-304.
[19] I. Kanev, D. Zaleski, B. Dave and W. Sanger, „Larger stalks and satellites of chromosome 21 inpatients with Down Syndrome,“ The Journal of the Association of Genetic Technologists, 2003, vol. 29(3), p. 116.
[20] I. Kanev, D. Zaleski, A.Z.Van Dyke, T. Hempel, M. Wiggins, A. Olney, B.J. Dave and W.G. Sanger, „A rare form of Down syndrome mosaicism and unusual parental cytogenetic characteristics,“ The Journal of the Association of Genetic Technologists, 2004, vol. 30 (3) p. 109.
[21] A. Anuradha, M. Satyanarayana and K. R. Manjunatha, „Satellite associations in recurrent aborters,“ The Indian for Human Genetic“, vol. 2(1), pp.61-64.
[22] K. Novak, D. Zaleski, M. Nipper, M. Wiggins, T. Hempel, I. Kanev, W. Sanger and B. Dave, „Mosaicism and isodicentric Yq in two phenotypic females and two phenotypic males,“. 2007, The Journal of the Association of Genetic Technologists, vol. 33(3), p. 154.





Накратко резюмето на професор д-р Иван Канев е следното:
Професионален учен с девет признати в Америка и Европа научни специалности.
За успешна съвместна работа по международни научни проекти е получил медали от:
Руската академия на науките.
Университета в Тел Авив.
До сега е публикувал над 200 научни статии с 130 учени от цял свят.
Номиниран е за Българин на годината в САЩ за 2007 година.
Има диплома „Златен орел“ на Българо-Американска Асоциация.
Съществува потенциална възможност след години тази идея да бъде предложена за Нобелова награда.